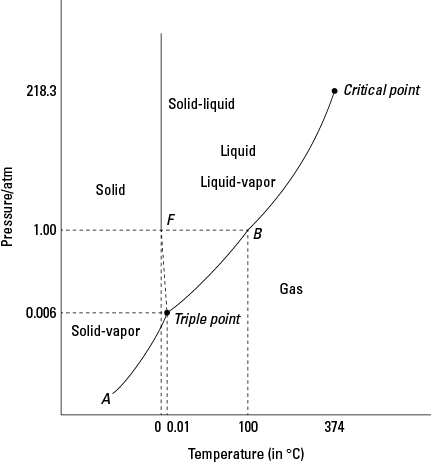
I’m not sure what is more geeky: my favorite chemistry graph itself or the fact that I have one. But seriously, the phase diagram is so elegant! In one picture, it explains how temperature and pressure relate to the states of matter. The lines are phase transitions (like condensation, sublimation, and solidification), interesting edges for investigation to understand how the particles behave.
What does this have to do with my reading? Steven Johnson, in his book Where Good Ideas Come From, draws from many examples of innovation across history and across disciplines, many of which are from science, but in particular he uses the analogy of “liquid networks.” In a liquid, there is enough structure for particles to mix and combine but enough energy for them to move around and slide past each other whereas in a solid they would just be stuck in place and in a gas they would collide and fly away. In his analogy, the particles in a liquid social network are ideas and people.
Continue reading “Book Notes & Thoughts: Where Good Ideas Come From”
You must be logged in to post a comment.